The circular sheets that Raffaele Mezzenga gently lifts from a petri dish are shiny and black. Looking at this tiny piece of paper, one could hardly imagine that it consists of a novel nanocomposite material, with some unprecedented and unique properties, developed in the laboratory of the ETH professor.
This new “paper” is made of alternating layers of protein and graphene. The two components can be mixed in varying compositions, brought into solution, and dried into thin sheets through a vacuum filter – “similarly as one usually does in the manufacture of normal paper from cellulose” says Mezzenga. “This combination of different materials with uncommon properties produces a novel nanocomposite with some major benefits,” says the ETH professor. For example, the material is entirely biodegradable.
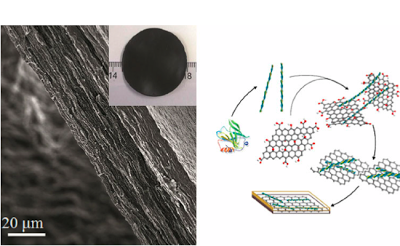
The final hybrid nanocomposite paper made of protein fibrils and graphene after vacuum filtration drying. The schematic route used by the researchers to combine graphene and protein fibrils into the new hybrid nanocomposite paper. (Reproduced from Li et al. Nature Nanotechnology 2012)
“Graphene paper” has shape memory features
Graphene is mechanically strong and electrically conductive, as well as, highly water repellent by nature. On the other hand, the protein fibrils are biologically active and can bind water. This allows the new material to absorb water and to change shape under varying humidity conditions. Furthermore, the “graphene paper” has shape memory features such that it can deform when adsorbing water, and recover the original shape upon drying. This could be used, for example, either in water sensors or humidity actuators.
But “the most interesting feature is that we can use this material as a biosensor to precisely measure the activity of enzymes,” says Mezzenga. Enzymes can digest and break down the protein fibrils. This changes the resistance of the composite, which is a measurable quantity once the graphene paper is incorporated into an electrical circuit. “This feature is, for me, the nicest part of the story. Seen from this angle, we could claim to have discovered a new general method to measure enzymatic activity”, says the ETH professor.
The material can also be designed to meet other needs. For example, the higher the proportion of graphene, the better it conducts electricity. On the other hand, the more fibrils are present, the more water can be absorbed by this material, with enhanced deformations in response to humidity changes.
Interestingly, this new material can be made with relatively simple means. The protein, in this case, beta-lactoglobulin, a milk protein, is first denatured by high temperatures in an acidic solution. The end-products of this denaturation process are protein fibrils suspended in water; these fibrils then act as stabilizers for the hydrophobic graphene sheets and allow them to be finely dispersed in water and processed into nanocomposites by a simple filtration technology.
The concept can be extended
In view of the widespread tendency of proteins to form fibrils, under specific conditions, this concept can be extended, in principle to other food proteins, such as those found in eggs, blood serum and soy. The beta-lactoglobulin fibrils used in the work lead by Mezzenga are digested specifically by pepsin, an enzyme present in the stomach to enable the digestion of several food components. However, varying the protein types could provide a new method of targeting a much larger class of enzymes.
Inspired by their past research on amyloid fibrils and by the rise of graphene, the ETH researchers have combined these two building blocks to generate a new class of versatile and functional materials. “Nowadays, graphene paper is no longer a novelty”, says Mezzenga, “it is the combination with amyloid fibrils which is central to this new class of hybrid materials”.
Graphene has exceptional mechanical and electronic properties but its hydrophobic nature is a disadvantage in biologically related applications. Amyloid fibrils are naturally occurring protein aggregates that are stable in solution or under highly hydrated conditions, have well-organized supramolecular structures and outstanding strength. Here, we show that graphene and amyloid fibrils can be combined to create a new class of biodegradable composite materials with adaptable properties. This new composite material is inexpensive, highly conductive and can be degraded by enzymes. Furthermore, it can reversibly change shape in response to variations in humidity, and can be used in the design of biosensors for quantifying the activity of enzymes. The properties of the composite can be fine-tuned by changing the graphene-to-amyloid ratio.
If you liked this article, please give it a quick review on ycombinator or StumbleUpon. Thanks

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.