Ads : Nano Technology Netbook Technology News Computer Software
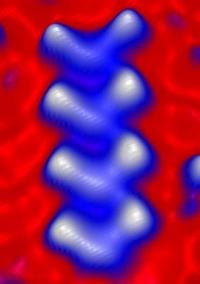
This image shows the smallest superconductor, which is only .87 nanometer wide.
Via Eurekalert Ohio University – Scientists discover world’s smallest superconductor
Scientists have discovered the world’s smallest superconductor, a sheet of four pairs of molecules less than one nanometer wide. The Ohio University-led study, published Sunday as an advance online publication in the journal Nature Nanotechnology, provides the first evidence that nanoscale molecular superconducting wires can be fabricated, which could be used for nanoscale electronic devices and energy applications. The study paves way for development of nanocircuits for energy, electronics applications.
Nature Nanotechnology – Superconductivity in just four pairs of (BETS)2GaCl4 molecules
This image shows several nanoscale superconducting molecular wires on a silver substrate.
How small can a sample of superconducting material be and still display superconductivity? This question is relevant to our fundamental understanding of superconductivity, and also to applications in nanoscale electronics, because Joule heating of interconnecting wires is a major problem in nanoscale devices. It has been shown that ultrathin layers of metal can display superconductivity but any limits on the size of superconducting systems remain a mystery. (BETS)2GaCl4, where BETS is bis(ethylenedithio)tetraselenafulvalene, is an organic superconductor, and in bulk it has a superconducting transition temperature Tc of ~8 K and a two-dimensional layered structure that is reminiscent of the high-Tc cuprate superconductors. Here, we use scanning tunnelling spectroscopy to show that a single layer of (BETS)2GaCl4 molecules on an Ag(111) surface displays a superconducting gap that increases exponentially with the length of the molecular chain. Moreover, we show that a superconducting gap can still be detected for just four pairs of (BETS)2GaCl4 molecules. Real-space spectroscopic images directly visualize the chains of BETS molecules as the origin of the superconductivity
4 pages of supplemental material
“Researchers have said that it’s almost impossible to make nanoscale interconnects using metallic conductors because the resistance increases as the size of wire becomes smaller. The nanowires become so hot that they can melt and destruct. That issue, Joule heating, has been a major barrier for making nanoscale devices a reality,” said lead author Saw-Wai Hla, an associate professor of physics and astronomy with Ohio University’s Nanoscale and Quantum Phenomena Institute.
Superconducting materials have an electrical resistance of zero, and so can carry large electrical currents without power dissipation or heat generation. Superconductivity was first discovered in 1911, and until recently, was considered a macroscopic phenomenon. The current finding suggests, however, that it exists at the molecular scale, which opens up a novel route for studying this phenomenon, Hla said. Superconductors currently are used in applications ranging from supercomputers to brain imaging devices
In the new study, which was funded by the U.S. Department of Energy, Hla’s team examined synthesized molecules of a type of organic salt, (BETS)2-GaCl4, placed on a surface of silver. Using scanning tunneling spectroscopy, the scientists observed superconductivity in molecular chains of various lengths. For chains below 50 nanometers in length, superconductivity decreased as the chains became shorter. However, the researchers were still able to observe the phenomenon in chains as small as four pairs of molecules, or 3.5 nanometers in length.
To observe superconductivity at this scale, the scientists needed to cool the molecules to a temperature of 10 Kelvin. Warmer temperatures reduced the activity. In future studies, scientists can test different types of materials that might be able to form nanoscale superconducting wires at higher temperatures, Hla said.
“But we’ve opened up a new way to understand this phenomenon, which could lead to new materials that could be engineered to work at higher temperatures,” he said.
The study also is noteworthy for providing evidence that superconducting organic salts can grow on a substrate material.
“This is also vital if one wants to fabricate nanoscale electronic circuits using organic molecules,” Hla added.
Advertising
Trading Futures
Nano Technology
Netbook Technology News
Computer Software
Future Predictions
Thank You

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.