The targeted removal of genes — the exact opposite of what a gene therapist would do — can restore cellular function in cells with genetic defects, such as mutations.
After the 2003 Northeast electrical power grid blackout, where a sequence of failures in the power grid led to the largest outage in U.S. history, experts determined that the event could have been reduced or avoided by instigating small intentional blackouts in the system during the initial hours of instability.
“And the same could be valid in biology, where a defective gene may trigger a cascade of ‘failures’ along the cellular network,” said Motter, whose interest and expertise lie in complex systems and understanding how the structure and dynamics of a high-dimensional system, such as an intracellular network or a power grid, relate to its function.

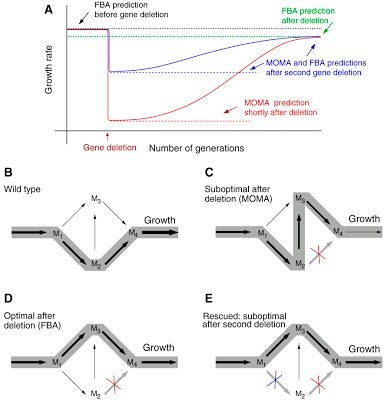
Schematic illustration of the consequences of gene deletion on the organism’s growth rate. (A) The growth rate following the deletion of an enzyme-encoding gene often drops, but after many generations may recover to a new optimal value not very different from the original one (red line). The optimal growth rate before and after the deletion is predicted by FBA (black and green dotted lines). The blue line indicates the predicted buffering effect of additional gene deletions: by deleting appropriately selected additional genes, the suboptimal growth rate shortly after gene deletions is higher than without the rescue deletions. (B–E) The effect of rescue deletions on the fluxes of a metabolic network, where M1 … M4 represent metabolites and the width of the arrows represents the strength of individual fluxes.
The team’s use of predictive models is similar to how physicists use models, for example, to determine the position of the moon tomorrow at a specific time. Thanks to the recent wealth of available biological information, computational scientists now are beginning to develop quantitative models of biological systems that allow them to predict cellular behavior.
In one in silico experiment (via computer simulation) with E. coli, the researchers found that the deletion of one gene is lethal to the cell but when that same gene is removed along with other genes, it is not lethal. The gene, it turns out, is only essential in the presence of other genes. This touches the issue, says Motter, of whether organisms have an unconditional set of essential genes.

Distribution of metabolic fluxes in the E. coli ‘s TCA cycle in arabinose minimal medium for (A) wild-type organism predicted by FBA, (B) fbaA mutant predicted by MOMA, (C) optimal state of fbaA mutant predicted by FBA, and (D) fbaA mutant with the rescue deletions of genes aceA and sucAB, predicted by MOMA. Key flux changes are highlighted in orange. Note that the metabolic flux pattern predicted by MOMA after the fbaA deletion (B) is similar to the wild-type fluxes (A). With the rescue deletions, however, MOMA-predicted fluxes (D) are brought closer to the FBA-predicted fluxes (C), restoring the organisms’ ability to produce biomass.
While Motter’s team has not done actual laboratory experiments, they have used their computational results to re-interpret and explain specific recent experimental results. They have applied physics methods to solve a biological problem. Their method, for example, can identify the genes whose removal restores growth in gene-deficient mutants of E. coli and S. cerevisiae, a type of yeast.

The impact of rescue deletions. (A) Predicted biomass production for the fbaA mutant of E. coli in arabinose minimal medium as a function of the number of rescue deletions when starting with aceA and sucAB. Deleted rescue genes are indicated in the figure. (B) Biomass production of tpiA- and nuoA-deficient mutants in glucose minimal medium as a function of the number of individual rescue deletions. Deleted genes are indicated in the figure. The optimal biomass flux remains unchanged with the addition of rescue deletions. The biomass fluxes are normalized by the wild-type flux GwtFBA=0.745 mmol/g DW-h in (A) and 0.908 mmol/g DW-h in (B).
FURTHER READING
The research is here: Predicting synthetic rescues in metabolic networks.
An important goal of medical research is to develop methods to recover the loss of cellular function due to mutations and other defects. Many approaches based on gene therapy aim to repair the defective gene or to insert genes with compensatory function. Here, we propose an alternative, network-based strategy that aims to restore biological function by forcing the cell to either bypass the functions affected by the defective gene, or to compensate for the lost function. Focusing on the metabolism of single-cell organisms, we computationally study mutants that lack an essential enzyme, and thus are unable to grow or have a significantly reduced growth rate. We show that several of these mutants can be turned into viable organisms through additional gene deletions that restore their growth rate. In a rather counterintuitive fashion, this is achieved via additional damage to the metabolic network. Using flux balance-based approaches, we identify a number of synthetically viable gene pairs, in which the removal of one enzyme-encoding gene results in a non-viable phenotype, while the deletion of a second enzyme-encoding gene rescues the organism. The systematic network-based identification of compensatory rescue effects may open new avenues for genetic interventions.

Brian Wang is a Futurist Thought Leader and a popular Science blogger with 1 million readers per month. His blog Nextbigfuture.com is ranked #1 Science News Blog. It covers many disruptive technology and trends including Space, Robotics, Artificial Intelligence, Medicine, Anti-aging Biotechnology, and Nanotechnology.
Known for identifying cutting edge technologies, he is currently a Co-Founder of a startup and fundraiser for high potential early-stage companies. He is the Head of Research for Allocations for deep technology investments and an Angel Investor at Space Angels.
A frequent speaker at corporations, he has been a TEDx speaker, a Singularity University speaker and guest at numerous interviews for radio and podcasts. He is open to public speaking and advising engagements.